
This article was originally featured on Hakai Magazine, an online publication about science and society in coastal ecosystems. Read more stories like this at hakaimagazine.com.
When a shark is born prematurely, salt water is toxic to its tiny body. A preemie baby shark can’t yet regulate the salt levels in its blood and will often die within a few hours. So researchers at the Okinawa Churaumi Aquarium in Japan came up with a solution: building an artificial uterus to simulate a mother shark’s womb.
In a new paper published in Frontiers in Fish Science, the researchers show that this manufactured uterus can sustain midterm Moller’s lanternshark embryos for up to a year, about two-thirds of their normal 18-month gestation period. The scientists hope this system will help sharks in their aquarium and eventually be used to bolster wild populations of other endangered shark species.
Lead author Taketeru Tomita had been studying shark and ray reproduction for several years when he joined the Okinawa Churaumi Aquarium and learned that sharks there occasionally give birth prematurely. The aquarium also receives living shark fetuses taken from mothers that die in fishing nets. This is a particular problem for viviparous sharks, which give birth to live young. Unlike oviparous sharks, which lay hardy egg cases, live-born sharks rely on the physical protection of the womb as they grow.
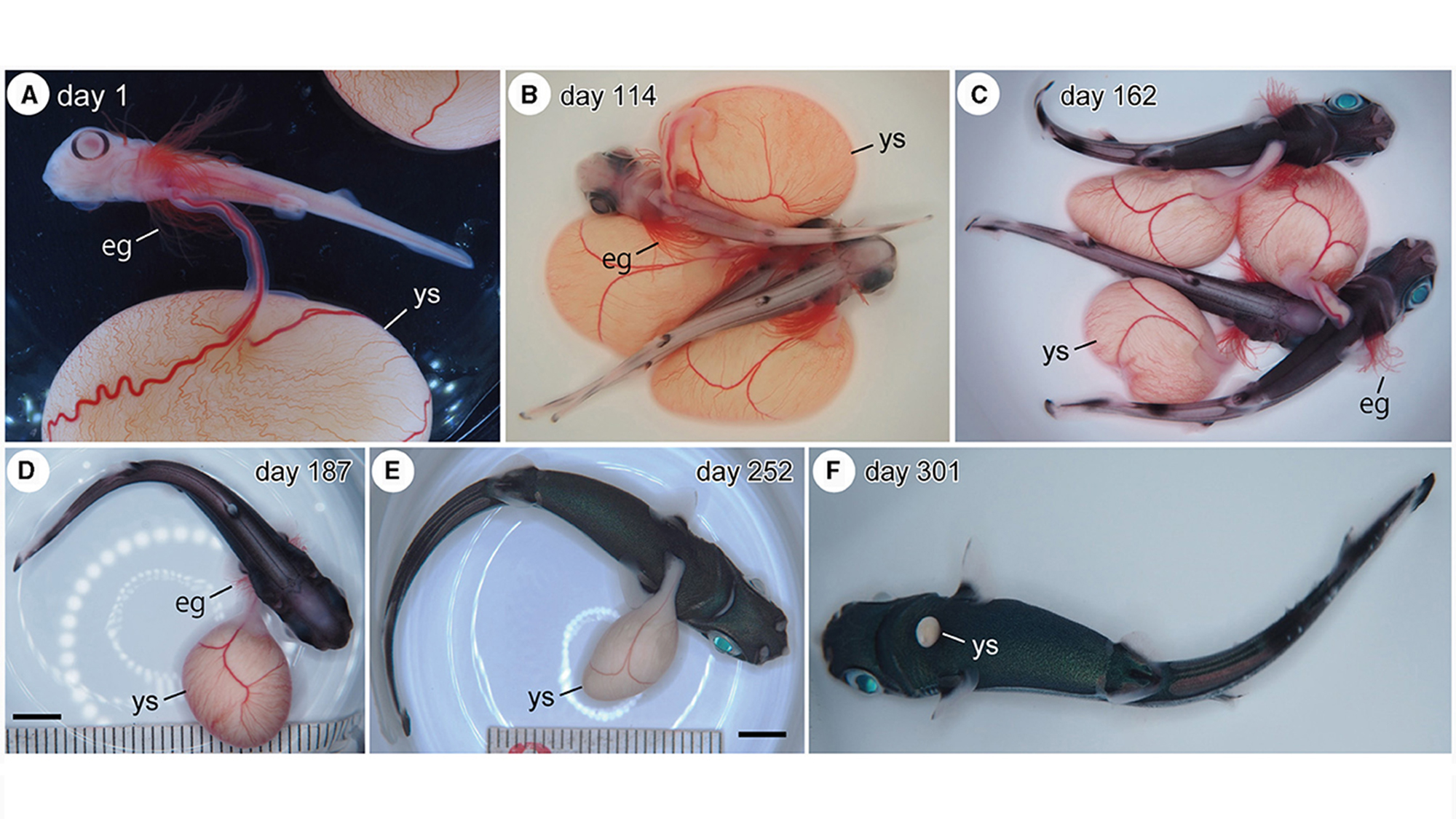
So Tomita assembled a team of shark scientists and the aquarium’s husbandry staff to see if they could instead build a uterus to give the preemies a little more time. The concept wasn’t entirely novel: shark researchers in Australia had previously gestated six ornate wobbegong pups for 18 days in an artificial uterus. For their version, the Okinawa Churaumi Aquarium team built a large tank; inside, they placed several smaller tanks to hold the growing embryos. Pumps circulated filtered, UV-sanitized fluid that mimicked sharks’ natural uterine fluid, and removed old fluid to be cleaned and reoxygenated.
This womb’s embryonic residents largely came to the aquarium from local fishermen, who sometimes accidentally catch pregnant Moller’s lanternsharks as by-catch and donate them to the aquarium if they die in the process. Luckily, because embryos of this species receive nutrients from external yolk sacs, they can survive for a short time after their mother’s death.
Still, the team’s early efforts were beset by challenges. One turned out to be the uterine fluid itself—specifically, figuring out how to wean growing sharks off it. In the first experiment, the team reared two late-term embryos for five months but watched them die when introduced to seawater shortly after “birth.”
In their second experiment, the researchers figured out how to slowly increase the proportion of seawater in the tank, mimicking how lanternsharks gently flush their wombs with seawater during pregnancy. Yet the team still lost many embryos to various complications—from an accidental spike in salinity to a problem with the oxygen supply to a suspected bacterial infection. Seven out of 30 survived.
“It was, of course, sad, but the more important thing in each moment was how to fix this problem,” Tomita says.
In their most recent effort, Tomita and his colleagues started with the youngest embryos yet—only about 60 to 80 days old. In the end, three out of 33 survived. Soon after being birthed from the artificial uterus, they enjoyed a birthday meal of minced mackerel. These sharks are now healthy adults, swimming in the Okinawa Churaumi Aquarium with no apparent differences from their peers.
Despite the sometimes grim business of playing shark midwife, Tomita is hopeful that these successes will translate into a tool that can be used for an even broader range of patients. Like many aquariums, the Okinawa Churaumi Aquarium receives donations of different shark species rescued from nets and beaches, which can be at different stages of pregnancy or have different reproductive strategies. In the future, they want to apply their efforts beyond lanternsharks and try to raise pups of other species, hopefully bolstering populations of the many sharks threatened with extinction.
“In our aquarium, as in the outside world for conservation, we cannot select the species we receive,” says Tomita. “The reproductive systems of sharks have great diversity, and I think our system can only apply to about half of viviparous sharks. We would like to develop more universal systems.”
Megan Ellis, a former shark researcher and colead of the Australian artificial uterus project, foresees this as another huge challenge. Although her team’s prototype incubated ornate wobbegong pups, it was intended for eventual use with the gray nurse shark, a critically endangered species in eastern Australia whose recovery is limited in part by its own reproductive strategy: pups cannibalize each other in the womb, reducing 40 embryos down to just two by birth. Ellis says preliminary research during their tests suggested that the ornate wobbegongs’ uterine fluid was completely different from that of gray nurse sharks.
Other sharks are more different still; great white sharks, for instance, provide additional nutrients through a milk-like fluid that seeps from the uterine wall. “That uterine fluid and the environment that an animal grows up in is going to be very species-specific,” Ellis says. “You might be able to do one fluid for a genus, but I don’t know whether you can make a uterine fluid that is one-size-fits-all.”
As for Tomita, his team’s next project will focus on sustaining lanternshark embryos through their entire 18-month gestation. He also wants to make the existing system more compact so that it’s easier to monitor over such a long period. “It was very tough work for one year. We had no weekends,” says Tomita of the most recent trial. “We always talked about how these sharks were like our own babies. They take a lot of work.”
This article first appeared in Hakai Magazine and is republished here with permission.
