
In November 2014, the Ebola outbreak in West Africa was at its peak. Thousands of people were dying in spite of public health strategies intended to limit its transmission. Officials at the World Health Organization (WHO) realized that people really needed a vaccine—scientists had developed one that seemed promising, but it needed to be evaluated quickly before it could be used to stop the outbreak.
How could researchers test the vaccine in Guinea, one of the countries most affected by the outbreak, with very little scientific infrastructure to help them? Several researchers presented their experimental protocol at the meeting of the AAAS on Friday, and discussed how it could be used to test possible treatments during other outbreaks, such as Zika virus.
Ebola spreads through families, neighborhoods and social circles, so the researchers decided to vaccinate people who had contact with those who had been infected. That protocol, called ring vaccination, was first used in the 1970s to eradicate smallpox.
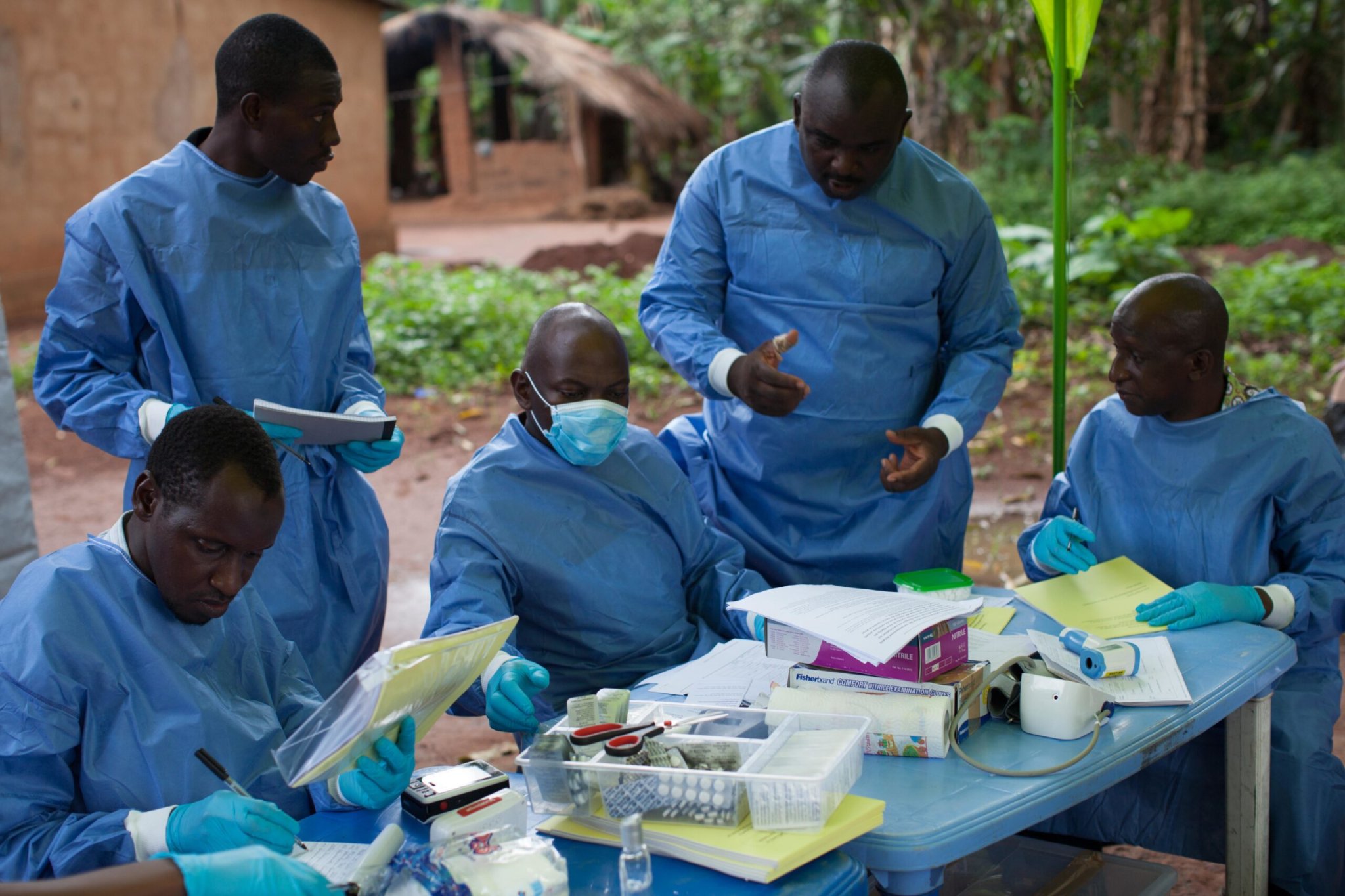
But this “surveillance and containment” strategy is difficult in countries with limited scientific infrastructure. To make the trial happen in Guinea, WHO officials organized a group of over 400 researchers and deployed them to 130 of the most affected communities in the country.
In each new community, the researchers asked permission from local leaders before starting the trial in order to reduce tension and build public trust. Then participants would sign forms indicating they understood the risks involved since the vaccine was experimental; those that couldn’t write their names signed with a fingerprint.

The most robust experiments compare the infection rate between people who received the vaccine and those who didn’t, but withholding the vaccine would be unethical in the midst of an outbreak like Ebola. So instead they vaccinated some participants shortly after a neighbor or family member was diagnosed, and then vaccinated the rest 21 days later. Each of the 7,600 participants was observed for 30 minutes after receiving the vaccine to make sure they didn’t have any adverse reactions. Then the researchers followed up six months later to see if the vaccine worked.
The researchers are still sifting through the data, but preliminary results suggest that the vaccine is 100 percent effective in preventing infection. And while this epidemic has largely abated because of public health strategies to stop the disease’s transmission, future outbreaks might be stopped more quickly with an effective vaccine.
Zika is much less deadly and harder to transmit than Ebola, but this vaccine test has taught public health officials some important lessons that might be used to combat it. The ring vaccination strategy could be used to test a Zika vaccine, too, since the disease also occurs in clusters as the mosquitoes that transmit the disease stay within a 1,300-foot range. Importantly, the researchers learned how to establish trust within a community that doesn’t have much scientific or government influence, which would be useful in remote regions in Latin America where Zika is rampant.
“This approach should work for Zika vaccine if we have one, and I think we will need one,” said Ira Longini, a statistician at the University of Florida who collaborated on the study. “Now, we want to make the point that we can almost certainly contain future Ebola outbreaks, and that we will probably have a new paradigm and tool for dealing with new outbreaks of whatever emerges in the future.”
The Ebola vaccine has not yet been approved, and testing it is a challenge since the outbreak has mostly abated. And while the researchers still have a few questions about how it works in the long-term, they are confident that they’ve found a potent tool to combat the virus: If another outbreak were to happen, Longini said, “I would want that vaccine. But you can look at the results and decide for yourself.”
With more cases of Zika virus diagnosed every day, hopefully a similar experimental strategy could help bring an effective vaccine to those who need it most.
