
Neurodevelopmental disorders like ADHD, autism, and other attention and learning disorders can all have different and unique symptoms that set them apart from each other. At the same time, though, people with these disorders seem to share one root symptom: They all struggle with something called sensory overload, in which too many distractions can lead to loss of attention, aggression, and trouble sleeping.
Treatments for these disorders exist, but none target the root cause, which scientists are just beginning to uncover. In a study published today in Nature, a group of researchers from New York University and MIT have identified a mechanism through which this sensory overload might arise. With it, the researchers think they can not only classify these disorders in a better way, but also develop therapies that alleviate or diminish the symptoms that make it impossible for some people who have them to function.
During sensory overload, too many distractions can lead to loss of attention, aggression, and trouble sleeping.
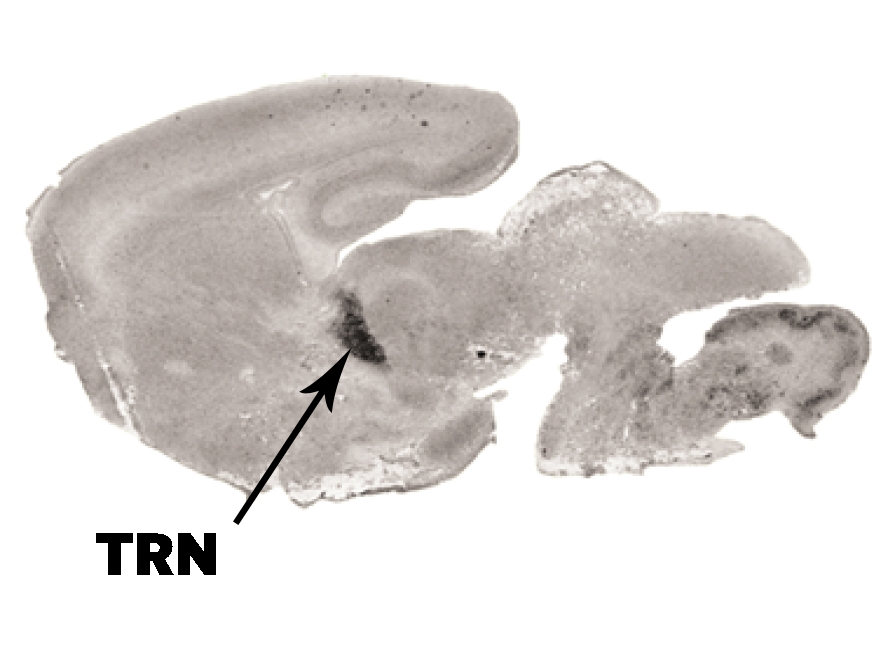
Back in October, Michael Halassa, a neuroscientist at New York University and a co-author of the new study, published a paper that found that a region of the brain called the thalamic reticular nucleus, or TRN, is responsible for controlling the brain’s ability to multitask. Acting as a task switchboard, the TRN processes sensory stimuli that come in, decides what’s most important, and directs attention to that. But for disorders like ADHD, autism, and schizophrenia, something within the TRN goes awry, making it harder, if not impossible for people with ADHD to focus on a particular task, or for people with autism to socialize, for example.
Halassa and his team, including Guoping Feng from MIT, wanted to know what was going on in the TRN to cause this. Previous studies had found that people born with defects in a gene that is most active in the TRN, called the PTCHD1 gene, struggle with sensory overload, leading to these cognitive conditions.
The researchers set out to understand exactly how that gene affects attention and distraction. Using genetically engineered mice that lack the PTCHD1 gene, they bombarded the mice with different flashes of light, only one of which meant a food reward. They also continually changed the flash of light that gave a food reward–forcing the mice to constantly alter their focus throughout the experiment. Compared to normal mice, mice that lacked the PTCHD1 gene made more errors, and their TRNs were less able to shut out the distractions to get the food reward–similar to how people who suffer from ADHD have trouble eliminating distractions to focus on the task at hand or how autistic individuals have a hard time tuning out what’s around them when having a conversation with someone.
“We clearly showed that the PTCH deletion from the TRN is sufficient to explain ADHD symptoms,” Halassa says. “Its this precise mapping, of circuitry to behavior,” he says, that makes him think they have nailed down a core symptom of these disorders, and what goes awry in the brain to cause it.
What Does This All Mean For Treating These Disorders?
Halassa thinks that this research could bring about a new way of categorizing these neurodevelopmental disorders like ADHD and autism. Currently, these conditions are identified individually, but if researchers could first pinpoint the core symptoms that cut across all these disorders (for example, sensory overload), then they may be better able to treat them.
“We think this is a general mechanism that can account for symptoms across these disorders,” says Halassa. Understanding this core deficit will better guide researchers to find ways to alleviate symptoms, he says.
Using their knowledge of the PTCHD1 gene, Halassa and his team were able to partially restore TRN signaling with an experimental drug tried out in the mice without the PTCHD1 gene. While it can’t be used in humans, it provides evidence that better knowledge of these pathways can give rise to new drugs that focus on getting the TRN back to normal functioning.
There’s still more work to do.
However they still have more work to do. Identifying other types of genetic abnormalities that give rise to sensory overload is an important next step. Then, Halassa says, they also need to perform imaging studies in order to understand how common sensory overload is across different disorders. Its also still unclear how big of an effect this sensory overload has on symptoms more specific to autism or ADHD. With this knowledge, they would know more exactly what effect restoring TRN functioning could have on each of the different conditions.
For now though, Halassa says, this study demonstrates what is perhaps a better way to approach their diagnosis and treatment: By focusing on what is similar, not different, to get at the root cause of a disorder.
Correction: An earlier version of this article originally referred to autism and ADHD as diseases, when in fact, they are neurodevelopmental disorders. The article has been updated to reflect this change.
