
The microbes in your gut depend on you to feed them well. A steady diet of complex fibers keeps them happy—and by extension, may keep you healthier. The microbiota have been linked to weight, gut health, allergies and even mood. Studies have shown that when the host (you) fails to supply the hungry hordes of beneficial microbes with what they want, the populations can change, and can even start to threaten the gut’s thin lining. But we have been unable to see exactly whether—and how—these shifts were happening.
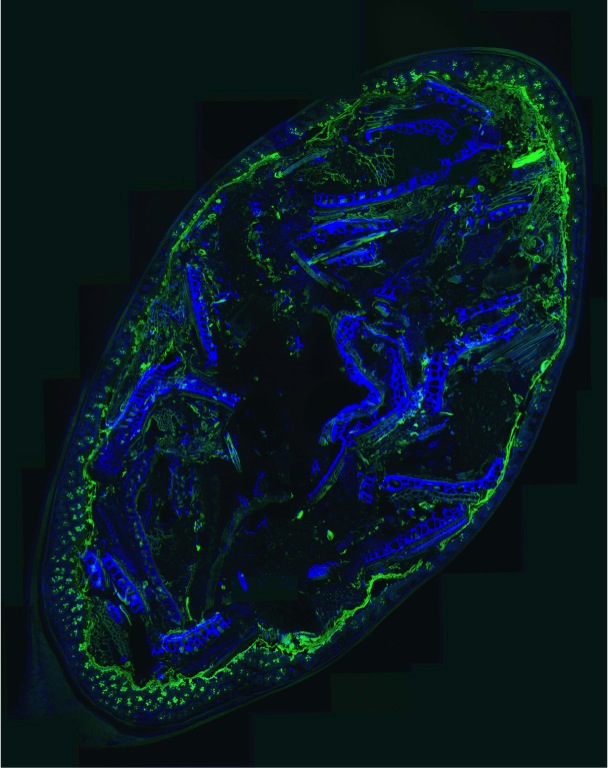
Now a team at Stanford University has created an elegant method of peering inside the gut—at cell-level resolution—to see what is going on. The research is in the October issue of Cell Host & Microbe.
Most of our understanding about gut microbes—and any impact diet has on them—has come from poop. Researchers can run a quick genetic scan on a smudge of a fecal sample, assessing which microbes are there, and in what abundances. But from this mixed-up pile, there is no way to know where in the gut the microbes are living—or how they are interacting with one another or with you. “Mapping the spatial organization of this microbial community is a fundamental aspect of understanding its biology,” says Justin Sonnenburg, a microbiologist at Stanford and coauthor of the new paper. “Without this information, we will struggle to make sense of how these microbes are contributing to our health or why interactions go awry and cause disease.”
“This is a huge advance,” says Eric Martens, an assistant professor of microbiology at the University of Michigan Medical School, who also studies the impact of fiber on gut microbes and the gut’s mucus layer and was not involved in the new research. “Since we are just beginning to understand the mechanisms and functions of the hundreds of species of bacteria that inhabit the gut, understanding how these organisms pack together, organize, and reproduce in such a dense and tightly confined space provides enormous insight,” he says.
Sonnenburg and his colleagues fed mice (colonized with human gut microbes) standard and fiber-deficient diets. They then carefully preserved thin slices of the mouse intestines and added special dyes to mark different microbes, undigested food, the essential mucus layer and the gut wall. With so many minute sample slices to analyze, they also developed software to help compute the spatial relationships.
In being able to actually see what was happening on these different diets, the researchers found that when the fiber was reduced, the mucus layer shrank—likely due to starved microbes eating it—from approximately 51 micrometers to just 31, allowing the microbes closer to the sensitive wall of the intestine. And as Sonnenburg notes, “we know that part of maintaining harmony between our resident microbes and our intestinal tissue is separation of the two”—namely by the mucus layer.
As this mucus layer shrank, mice missing the fiber in their diet also had more markers of inflammation. This can be triggered by the immune system attempting to keep bugs where they belong—in the gut. “Over long periods of time, low levels of inflammation can lead to many different types of problems, including colitis or even cancer,” Sonnenburg says. (But, he cautions, the experiments have only been in mice—and over a short time period—so how the findings translate to us and our long-term health remain to be seen.)
In addition to the shrinking mucus layer, the researchers were also able to see that changing the diet altered the way bacteria were organized in the gut. On a standard diet, two categories of bacteria were usually found in clumps of similar cells. But without fiber, these groupings vanished, and the microbes were more evenly distributed.
Ronan O’Connell, of University College Dublin’s Conway Institute of Biomolecular and Biomedical Research, has been using laser microscopy to parse the structure of the gut microbiota in healthy people as well as those with diseases of the intestinal tract. He notes that the new imaging technique “beautifully illustrates the complexity of the host-microbiome interface,” adding that it might some day be able to be used to study the differences in healthy and diseased guts in humans. Sonnenburg and his group are, in fact, already looking to expand the work to humans.
In the meantime, the new software used for this work (called BacSpace) is available to other groups, and, Sonneburg notes, he hopes sharing it will “propel this field forward rapidly.” Martens, for one, is enthusiastic about the potential for this visual approach. “One can imagine turning up the resolution to the species level, instead of just phyla.”
Katherine Harmon Courage is a freelance journalist; her next book Cultured (2016) is about the gut microbiota and diet. @KHCourage







