Artificial Wombs
Will we grow babies outside their mothers' bodies?

A fetus lives in a world of bubbles. In its earliest days, it’s shaped like one. Later, it floats in one-the squishy, enveloping amniotic sac. And eventually, if all goes well, the fetus releases one bubble of fluid, then another and another, like smoke signals, as it puckers and swallows and floats in the womb. It was the bubbles that first convinced Hung-Ching Liu two years ago that a baby might actually be grown outside its mother’s uterus. Liu, the director of the Reproductive Endocrine Laboratory at Cornell University’s Center for Reproductive Medicine and Infertility in Manhattan, has become, almost accidentally, the nation’s premier womb-maker.
Beginning in 2001, her lab started growing sheets of human tissue composed of cells from
the endometrium, the lining of the uterus. This engineered tissue, which used starter cells donated by infertile patients, was meant to bolster the clinic’s in-vitro fertilization success. A layer of endometrial cells is, after all, the ideal platform on which to nurture an embryo, a medium almost as good as mom would have made.
But the tissue, a single layer of cells stretched across a cell culture within a plastic petri dish, was gossamer-thin. “We’d hoped the embryos would implant on this tissue,” Liu says, “so we could learn more about the mechanisms of implantation. But they could not.” The growing embryos would break through the tissue, smack against the petri dish and, like a tree whose roots hit rock, die.
So Liu added layers, tissue atop tissue, until she had a three-dimensional model, essentially a freestanding uterus. Embryos could attach to this engineered tissue. They could burrow in, sending out shoots of blood vessel. They could take in nutrition and give out waste. They could divide, differentiate, and thrive.
Which led at last to the bubbles. In 2003, in an experiment that hasn’t received as much attention as one might expect–perhaps because Liu hasn’t published her results, due to her qualms about how those results will be received by politicians, activists and desperate would-be parents–a mouse embryo grew almost to full term in one of Liu’s artificial wombs. It moved. It breathed. It bubbled. “And not just one bubble,” Liu says. “We saw bubble, bubble, bubble.”
She may also have glimpsed, in that moment, the far-out future of human reproduction, vitreous and shifting. Thanks to her research and others’, man-made mouse wombs could be a reality within a decade–and a stepping stone to artificial human wombs. Eventually, these baby incubators could supplant natural ones. Conception could be clinical, and birth bloodless. Gestation could be detached from motherhood, and a fetus could be viable from the instant that sperm and egg fused.
Or not. Days after cheerfully percolating, Liu’s rodent fetus died, deformed and contorted, more seahorse than mouse, a developmental freak. The same thing happened to the next fetus she implanted, and the one after that. “Making babies is more complicated than we imagined,” Liu says. “And we knew going into this that making babies is very, very complicated.”
Liu, 63, is a short woman with full cheeks, a rounded middle and smooth, unlined skin who bustles along the corridors of her lab like a cartoon teapot. She is, by any measure, maternal, with two grown sons. Twenty-one years ago, Liu, then an assistant professor, joined the first team in the U.S. to have produced a test-tube baby. Now she and her partners can boast an enviable success rate. In the past several years, about 40 percent of the couples that came to her group conceived, and in 2004, the center’s 10,000th IVF baby was born.
Her artificial-womb work was a natural outgrowth of the IVF efforts and is motivated by the same ache. “I see so many women who want their own baby so badly,” she says. Among them are women whose embryos have failed to take hold and grow and who might benefit from her current research.
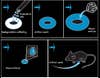
Liu’s artificial womb is a surprisingly simple construction. She created it after researching the making of artificial skin and adapting those methods. First she and her co-workers mold a base, a womb-shaped matrix of collagen and chondroitin, substances that are biodegradable. Over time, they dissolve, leaving only the endometrial tissue that is placed over the matrix. Each womb is shaped like a section of the mammalian version it mimics: The artificial human mold is bowl-shaped; the faux mouse womb is a doughnut-shaped section of a mouse’s tubular uterus.
In the beginning, Liu used endometrial cells donated by some of the clinic’s female patients to grow human tissue. Then she added human embryos left over from IVF treatments, donated by other patients. These zygotes implanted and started to grow. But after they had gestated for 10 days, Liu ended the experiments, well short of viability. Under current federal regulations, two weeks is the limit for human fetal growth in the lab. “So we switched to an animal model,” Liu says with a shrug. In 2002 she and her colleagues started making mouse wombs and growing mouse embryos inside them.
In outline, the gestation process seems straightforward. Sperm and egg meet. An embryo implants. Between them, mother and baby build a placenta and an amniotic sac. Fluid builds up around the growing embryo. Hormones move in and out. Nutrition, blood and oxygen pass through the placenta. Waste products are removed. There’s a gentle hum of maternal heartbeat and digestion. It’s like a well-modulated, high-end aquarium.
Except, of course, that it’s not. The actual sequence of events is exceedingly intricate. Miss one minor step, delete a gene expression, add a dribble too much or too little of a single hormone, and you’ll wind up with a baby who is dead or monstrous or, in what may be a blessing, both.
Artificial wombs have figured for generations in fiction, feminist theory, abortion debates and even the wistful imaginings of women far advanced in pregnancy. In Brave New World, the 1932 book by Aldous Huxley, babies grew in tubes. In 1970 Shulamith Firestone wrote in The Dialectic of Sex: The Case for Feminist Revolution that artificial wombs should be developed to free women from “the tyranny of their sexual-reproductive roles.” More recently, the exhausted working-mother heroine of the novel I Don’t Know How She Does It assures a younger female colleague that she, at least, will be relieved of the tedium and career-torching effects of pregnancy, thanks to emerging baby-in-a-box technology. Science has been playing catch-up to these fantasies: In 1963, researchers put miscarried fetuses in oxygen chambers and added a watery fluid. It didn’t work.
The closest approximation of an operational machine-womb was created about five years ago. In experiments at Juntendo University in Tokyo, an acrylic box was filled with a liquid similar to amniotic fluid. A goat embryo, removed by cesarean section after four months of normal gestation, was placed in the chamber and its umbilical cord hooked to tubes connected to an artificial placenta. Most of the kids died, but a few survived up to three weeks, reaching full term for a goat. None was without deformities or lung problems. The experiments are no longer under way.
Which has left the field to Liu. She and two of her colleagues, both men, have been refining their artificial womb bit by bit. They’re developing liquid formulas that are incrementally closer to the fluids within an actual mammalian uterus, although developing the perfect mix of blood, hormones and proteins–and precisely adjusting it during the course of a pregnancy–is so far impossible. Minuscule differences in amniotic fluid have been found, in other labs’ experiments, to produce notable differences in the resulting offspring.
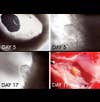
In Liu’s most recent experiment, she surgically implanted one of her artificial mouse wombs in an adult mouse, and the fetus inside lived about 19 days. (In earlier experiments, fetuses survived in external wombs for up to 17 days. That’s roughly equivalent to 37 weeks of human fetal development, although fetuses appear to develop slower in artificial wombs than in real ones.) But invariably, each mouse embryo, more than 150 to date, died. One reason, Liu believes, is that the snaky vines of blood vessels that should link the tiny bodies to the womb’s surface wither or fail to develop at all. Starved of blood, the embryos shrivel.
To rectify this, Liu’s colleague Weidong Wang has been studying the expression of a gene, called murine AGPAT, that seems to stimulate blood-vessel formation within the womb. If you block that gene, the embryo can’t implant fully and grow, which may explain why some women–in whom this gene is missing or malfunctioning–miscarry. Force the gene’s expression, on the other hand, and you get a jungle of blood vessels, a fecund clot of veins. The work may have implications for cancer treatment. Block the gene’s expression, and you could disable a tumor’s ability to create new blood vessels for itself, causing a slow self-strangulation. “There’s overlap between research into fertility and oncology,” Wang says. “The beginning of life and the end of life. We deal with both in this lab.”
The people most invested in the creation of an artificial womb may not be the scientists. Members of the Raelian cult–who also claim to have cloned a baby–announced in 2003 that they had developed a Babytron (their word) that could incubate human embryos from conception onward. No evidence exists to support either of the group’s audacious claims.
More seriously, some abortion foes, to support their fight, have seized on the potential of external gestation. As they point out, Roe v. Wade relies to a large degree on the nonviability of the aborted fetus. Forty states and the District of Columbia have restricted abortion after approximately 24 weeks of gestation, at which point a baby could survive birth. An artificial womb would vastly extend the period of fetal viability. In theory, an embryo could survive outside its mother’s body from the moment of its conception. Some anti-abortion activists have said that women should therefore be required to incubate aborted fetuses in artificial wombs.
Bioethicists worry that another subset of women will employ fake wombs for convenience, to avoid stretch marks and weight gain or to prolong Hollywood careers. Some radical feminists see the man-made uteruses as a way not just to free women from pregnancy but to rid the human race of females completely. If sufficient ova were banked, they say, men could have an artificial womb surgically implanted and bear children themselves.
Clearly, the most logical and worthy use of artificial wombs would be to help couples who cannot conceive but wish for a genetically related baby. But even in that case, the rationale for an artificial womb is murky. “We have a perfectly safe, workable alternative,” says Thomas Murray, the president of the Hastings Center, a bioethics think tank in Garrison, New York. “We have surrogate mothers.” An infertile woman might be squeamish about having someone else carry her issue, another body exchanging blood and emotion with her child. “But,” Murray continues, “if someone insists on an artificial womb, if they want to create a biologically related baby so badly that they are willing to risk having that baby be severely deformed, it’s hard for me to see the moral good in that.”
Janet DiPietro is the world’s leading expert on what it’s like to be an embryo. A developmental psychologist at Johns Hopkins University, she is one of the few scientists to have closely studied the womb as an ecosystem. In recent experiments, DiPietro and her colleagues attached monitors to the skin and belly of pregnant women. They had expected to find that the child within responded to its mother’s moods. Earlier research had shown that fetuses react almost instantly to changes in maternal position or emotions–with stillness. DiPietro speculates that this is the way the fetus learns to understand its mother and her body and how she reacts to noise and other stimuli.
But in work published last year, she found that the instruction is hardly unilateral. Fetuses teach their mothers, too. “We found that the fetuses were moving when the mothers weren’t even aware of it, and were giving the mother a little emotional jolt. They were, in effect, training her to pay attention to them.” Since much of this movement comes at night, they were also giving her a foretaste of sleep deprivation, apparently knowing deep within their DNA that she might as well start getting used to it.
“The fetal environment is more than just hormones,” DiPietro says. “You can’t simply add titrates of this or that hormone or protein and re-create the womb.” An embryo gestating outside its mother “will wind up being different than that same embryo [would be] had it developed the natural way.”
Women who have borne children understand this instinctively. Any woman who has lain in the dark watching a heel-shaped bump move across her belly knows that a sensibility is growing within, that the child is becoming itself even while still a part of her. The success of adoption shows that this interaction isn’t necessary for parental bonding. But is it essential in certain immeasurable ways to the infantile brain and body, to a baby’s later ability to touch, attach, and love?
As Liu pursues the science of hormone levels and gene expression, she too worries about the ineffable. In 2001, after her earliest experiments with human zygotes were publicized, she was inundated with calls from infertile women begging to become test subjects. Overwhelmed by the response and by her own unwonted realization that,
as she says, “this work could have great social impact,” she halted the artificial-womb experiments for a full year, resuming only after reaching certain decisions.
“I don’t want to make a womb for the convenience of women who don’t want to be pregnant,” she says. And she declines to discuss the uses that anti-abortion groups might make of her results. “I want to make a womb that would be a replacement organ,” she says, that would be implanted in a woman whose endometrial tissue was donated, that would fully re-create the rich, dark wilderness of a healthy female reproductive system.
But as we all know, intentions don’t mean much once an innovation is released. Liu thinks she and her team should have a viable mouse womb in 5 to 10 years. A human model will take longer–“10 years, maybe, or a little more,” she says, assuming that restrictions on fetal testing are lifted or eased. “It could take as much as 50 years, but I’m very hopeful that this is possible.” Her voice is soft. “It will be helping a life, a baby, helping parents. Those are good things, and that’s all I can be thinking about right now.”
Gretchen Reynolds lives in Santa Fe, New Mexico, and is mother to a son, Max. This is her first feature for Popular Science.
Baby’ first steps

Womb-Maker
Mouse Diary