
Proteins are like the workhorses of genetic biology, but they can be notoriously difficult to study. Their structure has everything to do with their function–and sometimes dysfunction–which has far-reaching implications in health and medicine. That’s why it’s such a big deal that a couple of researchers at Lawrence Berkeley National Laboratory have more or less hacked their cryo-electron microscope to see at far greater resolutions than its manufacturer intended and produced the first 3-D images of an individual protein with enough clarity to determine its structure.
Cataloging the shapes and structures of proteins is fairly routine science at this point. Pharmaceutical companies dealing in biologic drugs do so all the time as they search for protein therapies that might relieve one condition or another. But it’s not easy, and these conventional protein models are averages of the analyses of many thousands of molecules because it’s simply too difficult to get the resolutions necessary to image the features of an individual protein.
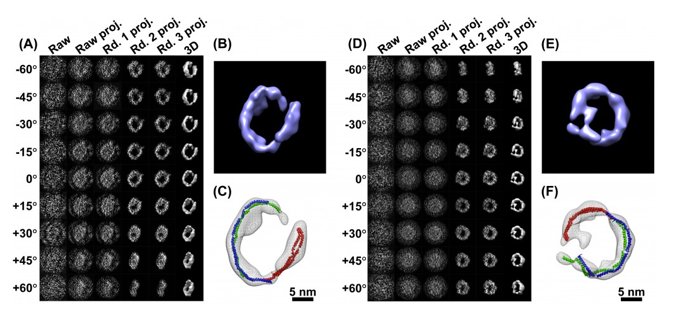
Until now. Gang Ren and Lei Zhang are reporting in the journal PLoS One the creation of their own brand of electron microscopy that they are calling “individual-particle electron tomography,” or IPET. Their images are still a bit fuzzy, but they are good enough for researchers to define a protein’s structure. Moreover, by creating a novel method of keeping their samples extremely cold (flash-frozen-in-liquid-nitrogen-to-negative-292-degrees cold) and tilting them up to 140 degrees while under the lens, they can generate more than a hundred images in a matter of a couple of hours.
Once stitched together those images inform each other, creating not only 3-D depth but helping to focus in on the subject protein and remove noise from the imagery. The result is the best structural imagery of an individual protein that we’ve ever heard about, one with the potential to go far in pharmaceutical research and in informing our fundamental understanding of protein dynamics. LBNL has a more in-depth explanation of the technology via the link below.
