Men lose Y chromosomes with age, and it might increase their risk of bladder cancer
The case of the missing Y chromosome and aggressive cancer growth is partially solved.
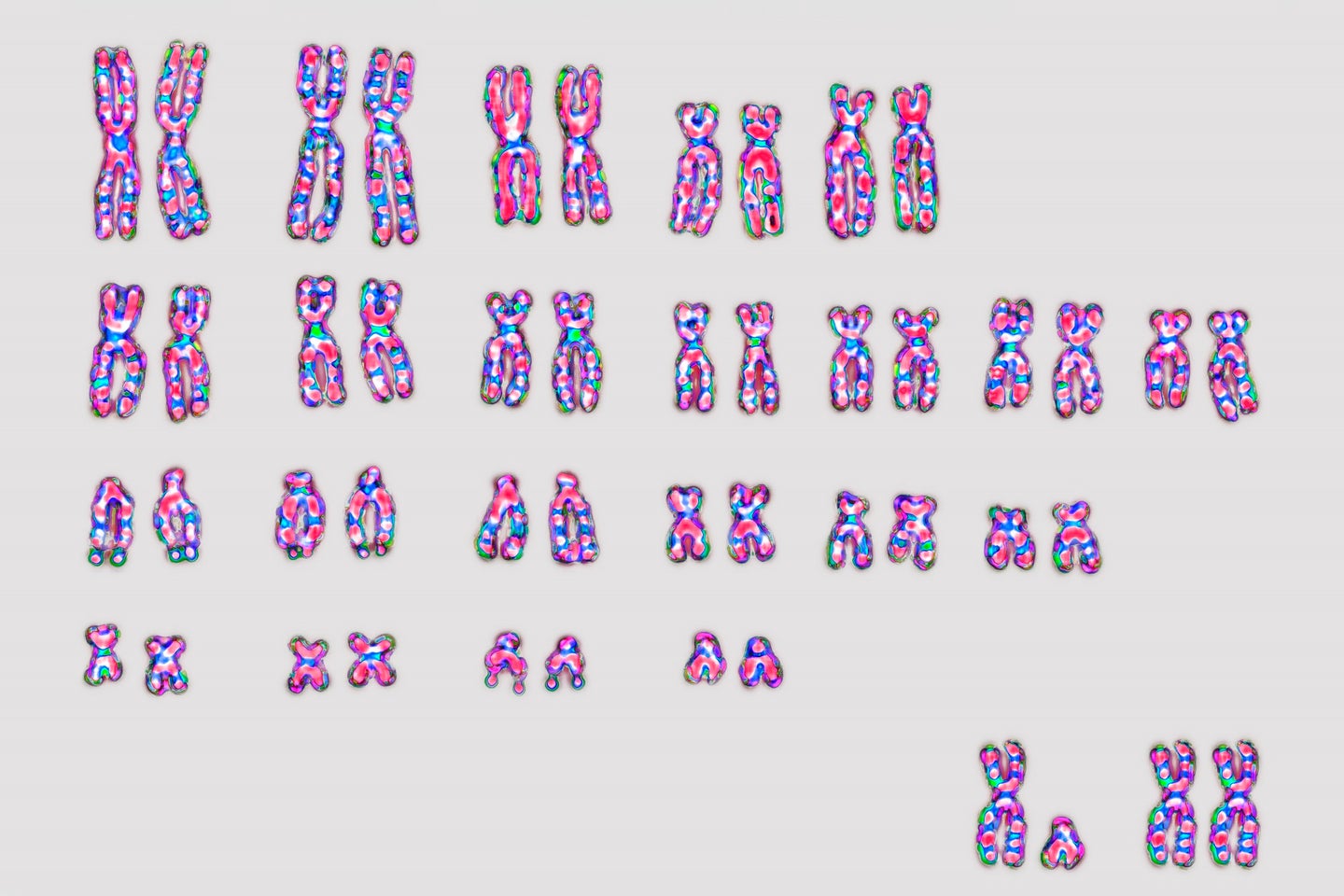
When urologic oncologist Dan Theodorescu thinks about his 25 years of research on bladder cancer, he remembers being in a flow. “When you hit something, you know in your gut that it’s really important and everything works out because you’re onto something.” His latest study, published on June 21 in the journal Nature, provides a clearer picture on why men have a greater risk for bladder cancer.
The trailblazing findings draw a clear connection between the disease and damage to the Y chromosome—the genetic structure that largely distinguishes male from female in mammals at birth. Bladder cancer is a sexually dimorphic disease with men making up a disproportionate amount of cases. In fact, men have a 1 in 28 chance of developing the cancer, compared to women, who have a 1 in 91 chance. Theodorescu, now the director of Cedars-Sinai Cancer, sought to understand why men are more prone to developing this type of cancer. Beyond risk factors like smoking cigarettes, he narrowed his decades-long search to changes in hormones and chromosomes. Previous work in the field had found that an increase in androgens, which are produced in the testes and ovaries, interferes with the body’s ability to fight off cancerous tumors. However, until this current study, few researchers focused on the role of chromosomes in bladder cancer.
As men get older, they naturally see the Y chromosome disappear in some cells. It’s a common phenomenon—at least 40 percent of men partially lose their Y chromosome by age 70. Previous research has linked that loss with several health problems, including an increase in heart failure, Alzheimer’s disease, and premature death.
[Related: What is a pangenome?]
Theodorescu and his team started by studying several bladder cancer cell lines (a population of cells bred from a single common cell). They grew and isolated two sets of cells from male mice whose fathers had either kept or lost the Y chromosome. Tumor cells without a Y chromosome were more likely to exhibit aggressive growth than those with the Y chromosome.
The next step involved pooling together 16 cancer cells from mice containing a normal Y chromosome with 16 cells missing a Y chromosome. Both cell lines had a similar growth rate. However, when they studied the same cell lines in animals, the authors noticed the tumor cells containing a Y chromosome did not grow as well as cells without a Y chromosome.
While the cell models hint at the sex chromosome’s role in cancer development, they don’t explain why it makes bladder tumors grow more aggressively. One hypothesis is that tumor cells without a Y chromosome grow faster because they have an easier time evading a person’s immune system. To test this possibility, the researchers injected Y-positive and Y-negative cells into mice bred without an immune system. Both tumor cells grew at the same rate, unlike in the cell lines where natural defenses were intact. “This was the clue that Y-negative cells were messing with the immune system somehow,” Theodorescu says.
Another experiment used genetically engineered mice missing various pieces of their immune system and found that immune cells responsible for fighting infection, called T cells, were the ones most affected by the loss of the Y chromosome. “We compared T cells from Y-negative to Y-positive tumors and found Y-negative tumors are doing things to T cells to get them exhausted,” Theodorescu explains. “An exhausted T cell can no longer destroy the tumor.”
The study might provide some insights on biological processes like adaptive immunity that are affected by disappearing Y chromosomes, says Chris Lau, a professor of medicine at the University of California, San Francisco who studies the human Y chromosome in his own research. “This could very well be the mechanism contributing to the disadvantage that seems to doom men with a mosaic loss of the Y chromosome in many other diseases, in which the immune system could play a role.”
Of course, the million-dollar question is whether doctors can use this knowledge to help patients with bladder cancer. One option includes immune checkpoint inhibitors. This common form of immunotherapy blocks the receptors that tumors use to send signals that confuse T cells. It also teaches T cells how to recognize cancer cells. When Theodorescu’s team administered immune checkpoint inhibitors to mice, they found those with Y-negative tumors responded better to treatment than Y-positive tumors.
[Related: A ‘living’ cancer drug helped two patients stay disease-free for a decade]
A new project Theodorescu and his colleagues are currently working on is seeing how many genes on the X chromosome are duplicates of genes found in the Y chromosome, also known as paralogs. Any mutations of these paralogs would cause a loss of gene function that could contribute to immune evasion in women, potentially adding to the risk of bladder cancer. Another issue the team is investigating is what happens to men who lose their Y chromosome and have mutations in Y-related genes on the X chromosome. “The loss may give you a worse cancer, but the response to immunotherapy could be even better,” Theodorescu notes.
If all goes well, Theodorescu says he plans to use the data from his recent work to create a test that would predict a person’s response to immunotherapy for any cancer. Additionally, the findings could help enhance immunotherapy and make it more effective against Y-negative tumors. Theodorescu is also not discounting the possibility of boosting immune response against other types of cancer. “If this pans out,” he says, “we may be in a situation where we could use this platform to discover combination therapies that could potentially cure a significant number of patients than checkpoint inhibitor therapy alone.”