The Essential Guide to Stem Cells
Everything you need to know about the hottest topic in medicine, from big-league breakthroughs and new therapies to emerging health risks and the patients willing to take them
For more than a decade, researchers have touted stem cells as the most promising advance in medicine since antibiotics. And this winter, when President Obama lifted the Bush administration’s ban on federal funding for embryonic-stem-cell research, talking heads buzzed that his decision could bring scientists that much closer to cures — not just treatments — for conditions like heart failure, spinal-cord injuries and Alzheimer’s disease. Biologists around the world toasted their new prospects with champagne. “Lifting the ban will free us up to use additional cell lines,” says Jack Kessler, director of the Feinberg Neuroscience Institute at Northwestern University. “It’s very important for science.”
The hype surrounding stem cells runs high these days. But getting the straight story — where the cells come from, what they do, and why they warrant executive orders and billions in research dollars — is surprisingly difficult. Making sense of the torrent of stem-cell research findings, separating the false claims from the scientists and studies that matter, requires an unusually well-honed baloney detector. In this comprehensive survey of the stem-cell landscape, we’ve done the vetting for you: hashing out the core science, analyzing the challenges, and getting firsthand insight from the patients themselves.
Stem Cells 101: Master the Lingo
Scientists talk up all types of stem cells and techniques to create them. But don’t feel overwhelmed — much of the jargon can be boiled down to these fundamental terms.
Embryonic stem cells: The Swiss Army knife of regenerative science, these cells are harvested in the early fetal stage and have the unique characteristic of pluripotency, meaning that they can turn into any one of more than 200 tissue types. This makes them ideal for regenerating diseased heart tissue, repairing spinal cords, and replenishing brain cells. But to critics who believe that human life begins at conception, harvesting these cells is akin to killing a baby.
Induced pluripotent stem (iPS) cells: These cells are as close as you’ll get to a fountain of youth. Inserting genes responsible for embryonic pluripotency into adult skin cells effectively rewinds their developmental clock and gives them embryonic-like powers to morph into heart, cardiac and other tissue types. An added bonus: No embryos necessary.
Somatic-cell nuclear transfer: This process birthed the famous cloned sheep Dolly. The basics: Take an egg cell and replace its nucleus with the genetic material of an adult cell from the organism to be cloned. Shocking the cell yields an embryo with the same DNA as the donor, which eliminates the risk of an immune reaction. But cloning humans may carry too much ethical baggage to be truly worthwhile, especially given the viability of iPS cells, which also contain a patient’s own DNA.
Cord-blood stem cells: These multipotent stem cells are derived from babies’ umbilical cords. Most of them are precursors for blood and immune cells, so they aren’t as versatile as embryonic or induced pluripotent adult cells. Recently, however, cord-blood stem-cell transplants have become a viable alternative to bone-marrow transplants in treating blood disorders like leukemia, especially when a bone-marrow match can’t be found.
Big Money, Big Picture
Ground Zero
This year delivers a major spike in federal funding for all types of stem cells, and new legislation puts fewer restrictions on how to spend it. With just one U.S. clinical trial of embryonic-stem-cell therapy under way, the hope is that this double shot of adrenaline will help American researchers pick up the pace.
Amount of money the National Institutes of Health (NIH) spent on stem-cell research in 2008: $938 million
Approximate amount of money the state-funded California Institute for Regenerative Medicine has approved in research grants since its inception in 2005: $693 million
Number of clinical trials on embryonic stem cells sanctioned by the Food and Drug Administration: 1
Estimated number of sanctioned clinical trials involving non-embryonic stem cells: 2,450
Estimated number of companies developing stem-cell products worldwide: 300
Approximate number of patients in the U.S. who have received stem-cell-based therapy within the past three years: 30,000
Estimated amount of annual revenue stem-cell therapies are expected to generate in the U.S. by 2018: $8 billion
Percentage of Americans in favor of embryonic-stem-cell research: 73
Sources: California Institute for Regenerative Medicine; clinicaltrials.gov; Geron Corporation, National Institutes of Health; RRY Publications; Time Poll, 2008
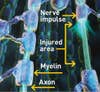
Nerve Fibers in the Spine
The Study to Watch
All eyes are on the world’s first human clinical trial of embryonic stem cells
Hans Keirstead will always remember the day he set down a once-paralyzed rat and watched it walk away. Keirstead, a biologist at the University of California at Irvine, along with his team at Geron Corporation in Menlo Park, California, were investigating a possible way to treat spinal-cord injuries with embryonic stem cells and, before that moment, not a single person in the lab knew which crippled rats had received the stem cells and which had not. “I’ll never forget holding the animals and knowing, without anyone telling me, which ones had been treated,” he says. “One of my grad students yelled, ‘We broke the code!’ It was so obvious. Some of the animals were walking, and some were not.”
But Keirstead hopes he’ll someday look back on this, the most transformative experience of his life so far, as a prelude to something much greater. In January the Food and Drug Administration gave Keirstead and Geron the green light to perform the same treatment again — only in paralyzed human patients. As the first U.S. clinical trial of embryonic-stem-cell therapy, Geron’s experiment represents what stem-cell biologists have so long been seeking: a chance to back up breezy hopes with concrete, dramatic results.
Like the rats in Keirstead’s earlier study, Geron’s human participants will receive injections of GRNOPC1, the company’s proprietary mixture of cells derived from embryonic stem cells. The compound is designed to work on spinal-cord patients with acute injuries. That means all the participants in the initial safety trial, slated to begin next month, will have been hurt within two weeks of treatment. In addition to improved walking abilities, rats that received the treatment within days of being injured showed significant healing of the spinal cord’s protective covering, myelin. Since Geron has submitted to FDA scrutiny for years, some scientists say the time is right to see if human patients can make the same strides as their animal counterparts. “This field needs a big success,” says Robert Lanza, the chief scientific officer of Advanced Cell Technology in Santa Monica, California. “The FDA is very careful. They would never allow clinical trials of a therapy like this unless there was proof it was safe.”
But critics worry that it could put patients’ health in danger, and that a botched trial could sour the public on future stem-cell treatments. “I have significant concerns that the spinal cord is not the best place to use stem cells for the first time. It’s a very complex environment,” says stem-cell expert Jack Kessler. “Everyone will be watching. If the trial doesn’t work, people will lose faith.” Keirstead acknowledges the risks, such as uncontrolled tumor growth, but feels compelled to press forward. “We’ve done everything we possibly can to ensure safety. Every scientist has to strike a delicate balance between scientific diligence and sympathy for patients’ desperate needs, and I feel we’ve struck that balance very well.” He’s looking forward to greeting treated patients in the clinic for the first time — and maybe, in his wildest dreams, watching them walk away.
How It Works
Inside the first government-sanctioned clinical trial to heal injured spinal cords with stem cells
A blunt blow or disease can cause paralysis by destroying nerve fibers, or axons, in the spinal cord that carry signals to and from the brain. When these signals are disrupted, the protective insulation surrounding the axons, known as myelin, erodes, leaving permanent damage. Starting next month, scientists at Geron Corporation will begin injecting myelin-precursor cells derived from embryonic stem cells into patients with newly injured spines. The hope is that the new cells will create myelin and restore the flow of nerve impulses traveling along the axons.
Patient Diary: Claudia Castillo

Clinical Trial
In 2008, Claudia Castillo, age 30, underwent an operation to repair her windpipe. To eliminate the risk of rejection, surgeon Paolo Macchiarini of the Hospital Clinic of Barcelona in Spain implanted a section of trachea seeded with stem cells taken from Castillo’s bone marrow. It was the world’s first transplant surgery involving stem cells.
Here’s what she says about the experience:
I was diagnosed with tuberculosis in 2004, and it gave me a lot of problems, especially at night. I kept coughing and coughing and getting worse. I spent a lot of time incapacitated, because I had to be quarantined. I also had bronchial stenosis, a condition in which the bronchial tubes narrow and make it hard to breathe. It’s hard to fix because you can’t just take out one section of the bronchus and leave the other. But my doctor said he had this new technique to transplant a section of trachea into the bronchus.
I was scared going into the operation. When you’re the first person in the entire world to have a procedure, even the doctors don’t know exactly what will happen. I walked into the operating room thinking, “Will I wake up? Will I not wake up?” They took a section of trachea out of the donor, cleaned it, and put my stem cells on it so they could implant it into me.
So far, my body hasn’t rejected the organ, and I’m feeling good. Now I can walk up stairs without having to stop after every two steps. There is still a lot of recovery time ahead of me, and I’m not like I was before the illness, but if other people are considering this surgery, I would say “Go for it!”
– As told to Tetsuhiko Endo
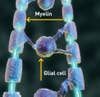
Myelin and Glial Cell
Brilliant Breakthroughs
These landmark studies deliver on the promise of stem cells and bring real therapies within reach**
Reversed Stroke Damage in Rats
Milestone: This 2008 study conducted by Gary Steinberg and his colleagues at Stanford University marks the first time researchers used embryonic stem cells to create mature brain cells that significantly improved an injured rat’s coordination, without causing tumors. When the researchers transplanted freshly grown neurons into rats that had suffered strokes and lost the use of one front paw, the rats regained control over the limb within two months.
Why It Matters: In the next few years, Steinberg hopes to adapt the technique to restore brain function in people who have suffered strokes and other neurological ailments.
Endowed Adult Stem Cells with Embryonic-like Powers
Milestone:In 2007, Shinya Yamanaka of Kyoto University in Japan and James Thomson of the University of Wisconsin announced almost simultaneously that they had successfully transformed mature skin cells into multipotent powerhouses that could morph into dozens of tissue types — a first in the field.
Why It Matters: Creating stem cells from a patient’s own tissue eliminates problems with immune rejection and sidesteps the controversial use of embryos, making them virtually ideal to treat disorders from diabetes to Alzheimer’s. But their inability to morph into as many tissue types as real embryonic cells remains a limitation.
Created Adult-stem-cell Lines for 10 Human Diseases
Milestone: Frustrated by the shortage of embryonic-stem-cell lines (limited to 22 under the Bush administration), last year George Daley and other scientists at the Harvard Stem Cell Institute took adult skin cells from patients suffering from a variety of conditions, including Parkinson’s disease and diabetes, and turned them into undifferentiated cells that behave like embryonic stem cells.
Why It Matters: These new stem cells could give rise to a limitless supply of tissues that could be used to study diseases and test medications before experimenting on animals and humans.
Cloned Human Embryos
Milestone: Robert Lanza and his team at Advanced Cell Technology have made significant progress toward therapeutic cloning. By removing the nuclei from human egg cells and replacing them with genetic material from adult cells, the researchers proved for the first time that the resulting cloned embryos were as healthy as normal embryos, adding weight to the argument that such clones could be viable sources of embryonic stem cells.
Why It Matters: Deriving embryonic stem cells from a patient’s own genes provides scientists with a plentiful source of patient-tailored cells and gets us one step closer to a new era of personalized medicine.
What Could Possibly Go Wrong?
For all their promise, stem cells harbor a dark side: Tumors
Sealed Fate
It started with a worst-case scenario. An Israeli boy was born with a rare disease called ataxia telangiectasia, which consumes parts of the brain and can cause paralysis. Anxious to halt the disease before the damage potentially turned lethal, the boy’s family sent him to a clinic in Moscow that gave him injections of neural stem cells from fetuses — once when he was 9 and then again when he was 10 and 12.
A year after the boy’s final treatment, disaster struck. He began complaining of headaches, and scans revealed that he had developed two nonmalignant tumors, one on his brain stem and the other on his spinal cord. Although he eventually underwent successful surgery to have the spinal-cord tumor removed, the case forced experts to reexamine the inherent risks of stem-cell treatment, particularly the tendency of transplanted stem cells to divide out of control. In a report on the Moscow debacle, Ninette Amariglio of the Sheba Medical Center in Israel wrote that mixing stem cells from multiple fetuses with growth-promoting compounds “may have created a high-risk situation where abnormal growth of more than one cell occurred.”
Other kinds of stem cells could have similar effects in humans if they are not properly directed to evolve into distinct tissue types, warns Jack Kessler of the Feinberg Neuroscience Institute. “If you take a human embryonic stem cell and transplant it into a person without the cell being differentiated, it will cause a tumor, period. We have to remove all possibility of that happening.”
Making sure tumors don’t develop in stem-cell recipients is a tall order, requiring researchers to finely control the growth of the transplanted stem cells. Too little control, and the cells divide willy-nilly; too much, and they lose their regenerative capacity. Researchers have recently learned more about the biological factors that control stem-cell proliferation — last year, Jurgen Knoblich of the Institute of Molecular Biotechnology isolated several cell proteins that can regulate or halt cell division. Still, the difficulty of striking the elusive cellular balance between stability and clinical effectiveness helps explain why we haven’t seen more stem-cell therapies in human trials yet. “I know there’s a lot of public pressure to demonstrate that stem-cell science is producing something,” says Marius Wernig, who directs a laboratory at the Stanford University School of Medicine, “but things can really backfire if we proceed too quickly.”
Quack-Science Checklist
Stem cells give new hope to thousands of people with chronic medical conditions, but shady researchers and clinics prey on that hope by fabricating results and offering untested treatments. Here are some hallmarks of sham science.
Sweeping promises
Chinese surgeon Hongyun Huang lured hundreds of desperate paralyzed patients to his Beijing clinic with claims that his treatments would provide “neuro-regeneration, repair and functional recovery.” But when a team of doctors investigated Huang’s work in 2006, they discovered that none of the spinal-cord-injury patients they followed showed any benefits from his therapy, and five suffered dangerous side effects. One man returned home with holes through his skull — Huang had placed cells in the man’s brain instead of his spine.
Blame-shifting
After an independent investigation in 2005 found that Korean researcher Hwang Woo-Suk had fabricated 11 of his stem-cell lines, Hwang held a press conference to apologize but still refused to admit that he had cheated. Rather, he blamed his collaborators for cooking up fake data and charged that they were involved in a conspiracy to sabotage his projects.
Highfalutin jargon
At the Cancun Stem Cell Clinic in Mexico, patients receive treatments from a “Hypoxicator” and a “Turbosonic Machine.” Without offering any evidence, the clinic’s Web site claims that these devices “stimulate your body to produce stem cells.”
Police tape
In 2006, the Dutch government shut down the PMC stem-cell clinic in Rotterdam after one patient was hospitalized for a serious allergic reaction to an unproven treatment.
Human Embryonic Stem Cell
What’s Next?
Stem cells: future cures and controversies
Landmark legislation to lift the ban on federal funds for embryonic stem cells, $10 billion in stimulus money, the first embryonic-stem-cell trial under way in the U.S. — it all adds up to a banner year for stem-cell science. But in a larger sense, things are just getting started. The next decade will see human trials of stem cells designed to treat all manner of illnesses and injuries, from blindness to diabetes, heart failure to paralysis.
More immediately, Advanced Cell Technology is slated to begin a human trial later this year that involves implanting embryonic stem cells into retinas to halt macular degeneration. Neuralstem in Rockville, Maryland, is lobbying the FDA for permission to start a trial using neural stem cells to treat ALS (amyotrophic lateral sclerosis, also called Lou Gehrig’s disease). And San Diego–based Novocell is developing insulin-producing cells from embryonic stem cells and hopes to receive permission to implant the cells in diabetic patients in the next few years.
As the number of embryonic-stem-cell trials increases, the number of paperwork headaches that go along with them will decrease, since reversing the ban effectively eliminates the artificial division between resources used for federally funded stem-cell research and embryonic-stem-cell research. Before, “having to exclude public funds from certain projects was a nightmare,” says Stanford University stem-cell expert Marius Wernig. As a result, we can expect to see the creation of new research-ready embryonic-stem-cell lines that carry disease-causing genes. These will help scientists study the origins of disease on a cellular level and test new medicines before experimenting on humans.
But as research on embryonic stem cells intensifies, so too will the ethical objections. “A lot of social conservatives say that destroying any embryo is wrong, and they will continue to be a very solid minority,” says David Masci of the Pew Forum on Religion and Public Life. Those objections could quiet down if researchers learn to work with adult stem cells, which don’t involve embryos at all. Cardiologist Eduardo Marban of the Cedars-Sinai Heart Institute in Los Angeles began a clinical trial this year that uses adult stem cells to repair cardiac tissue after a heart attack, and Joanne Hertzberg of Duke University plans to expand the cerebral-palsy trial in which Dallas Hextell participated, eventually offering the cord-blood stem-cell treatment to 100 children.
Although adult stem cells are promising, the need to avoid controversy can’t drive things — the field needs all the research options it can get. “There’s still so much we don’t understand, so we have no idea what type of stem cell is going to be best for which particular disease,” says Advanced Cell Technology’s Robert Lanza. “I think there’s a consensus in the scientific community that we need to pursue all of the avenues available.”
Patient Diary Dallas Hextell

More to See
In July 2007, Cynthia and Derak Hextell of Sacramento, California, enrolled their one-year-old son, Dallas, in a Duke University clinical trial to receive cord-blood stem-cell therapy for cerebral palsy, an incurable neurological disorder that affects muscle coordination. The hope is that the stem cells will repair the damaged tissues in Dallas’s brain. The first-of-its-kind trial, which will eventually enroll 100 children, is still in progress. Kurtzberg aims to publish her results next year.
Cynthia: Dallas was diagnosed with cerebral palsy when he was around eight months old. He was missing some milestones, like rolling over, and he cried constantly. After the shock of the diagnosis faded, I remembered that we had banked his cord blood after he was born, as a form of insurance, and I started doing some research.
Derak: The Duke treatment was simple. They just put the IV into his arm. It took 15 or 20 minutes for all the stem cells to get into his body.
Cynthia: Dallas started improving within weeks. It had always seemed like there was a fog over him, and he just started focusing better. It was like his brain was becoming more of a sponge.
Derak: He couldn’t crawl before we went to Duke, but now he’s walking and running.
Cynthia: Everything points to the fact that the stem cells worked, but it would be irresponsible to say either way until there is hard proof. We don’t think Dallas would have made the progress he made as quickly as he did without the stem cells. Now we have to wait for the trial results.