5-Minute Project: Hand Warmers
Make your own portable hot packs
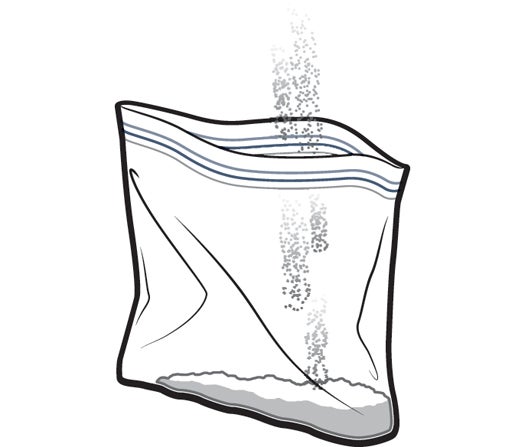

Instant hand warmers are great–just shake ’em up, and you’ve got spontaneous warmth to thaw your hands during the cold winter months. But they’re awfully expensive, and not because they’re complicated to manufacture. In fact, you can make them yourself in a few very easy steps.
Materials
- Calcium chloride ice-melt pellets
- Water
- Two ziploc bags of different sizes
Instructions
1. Fill a large ziplock bag one quarter of the way full with calcium chloride ice-melt pellets (available at most hardware stores).
Hand Warmers 1
2. Fill a smaller ziplock bag halfway with water, close tight, and place inside the first bag.

Hand Warmers 2
3. Squeeze the smaller bag until it breaks open, to create a heat-producing reaction that will last 20 to 60 minutes.
Hand Warmers 3
Originally posted by Justin DiPlacido on Instructables.